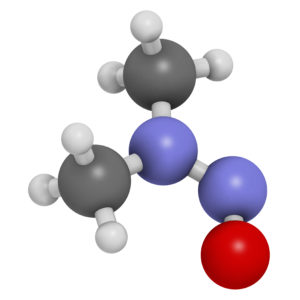
 The chemical N-Nitrosodimethylamine (NDMA) is a volatile, combustible, yellow, oily liquid that emits toxic fumes of nitrogen oxide once it is heated. NDMA is primarily used in laboratory research as a method of inducing tumors in rats. The substance can also be formed in foods containing sodium nitrite as a preservative, such as cured meats and fish. NDMA can also be found in some alcoholic beverages, cheeses, vegetables, smoked and pickled foods, sausages, beers, and fruits, and is a known contaminant in rubber products. NDMA exposure can lead to liver damage and irritation to the eyes and skin. NDMA can reasonably be anticipated to be a human carcinogen.
The chemical N-Nitrosodimethylamine (NDMA) is a volatile, combustible, yellow, oily liquid that emits toxic fumes of nitrogen oxide once it is heated. NDMA is primarily used in laboratory research as a method of inducing tumors in rats. The substance can also be formed in foods containing sodium nitrite as a preservative, such as cured meats and fish. NDMA can also be found in some alcoholic beverages, cheeses, vegetables, smoked and pickled foods, sausages, beers, and fruits, and is a known contaminant in rubber products. NDMA exposure can lead to liver damage and irritation to the eyes and skin. NDMA can reasonably be anticipated to be a human carcinogen.
NDMA and Other Nitrosamines Among Potent Group of Carcinogens
According to NCBI, nitrosamines are one of the most potent groups of carcinogens, with about 300 of the compounds tested, and about 90 percent being found carcinogenic in laboratory animals. Volatile NDMA has been shown to potentially induce tumors of the lung, bladder, kidney, liver, pancreas, esophagus, and tongue. Unfortunately, over the past year and a half, nitrosamines have been found in some blood pressure medications.
Science Transitional Magazine discusses the story of the chemical contamination found in generic blood pressure medications manufactured in China. Originally, there were two generic companies whose tablets included NDMA—Teva and Prinston. The Teva contaminated material was sold by Major Pharmaceuticals and Actavis, while the Prinston tablets were sold by Solco Healthcare. The issue became even more confusing because some of the tablets from those companies were fine because they came from a different manufacturer.
Generic Blood Pressure Medications Tainted with Nitrosamines
 Once the FDA began testing valsartan, losartan, and irbesartan in the generic forms, it was found that valsartan manufactured by Hetero Labs of India was also contaminated by NDMA. In late October, yet another drug in the same class of generic blood pressure drugs was found to be contaminated with NDEA, another nitrosamine. Like the NDMA-contaminated blood pressure drugs, the contaminated drug was repackaged under other names. Mylan pulled several lots of valsartan tablets which they had made themselves, as did Teva, Torrent, and Aurobindo, who had all bought some of Mylan’s tainted tablets.
Once the FDA began testing valsartan, losartan, and irbesartan in the generic forms, it was found that valsartan manufactured by Hetero Labs of India was also contaminated by NDMA. In late October, yet another drug in the same class of generic blood pressure drugs was found to be contaminated with NDEA, another nitrosamine. Like the NDMA-contaminated blood pressure drugs, the contaminated drug was repackaged under other names. Mylan pulled several lots of valsartan tablets which they had made themselves, as did Teva, Torrent, and Aurobindo, who had all bought some of Mylan’s tainted tablets.
More Blood Pressure Medications Recalled
As nearly as can be ascertained, there appears to be an issue with the actual process of the recalled blood pressure medications, which is assumed to be related to changes in the manufacturing conditions. Patients who take drugs containing the substance Valsartan have received notification of voluntary recalls of the tainted medications, due to the presence of NDMA. As time has passed, the problem continues to snowball, as more and more generic blood pressure medications have been found to have NDMA.
Lawsuit Filed in New Federal Court Targeting Companies Who Sold Tainted Valsartan
Drugwatch recently discussed a newly filed class-action claim which targets companies that distributed or sold valsartan, losartan, and irbesartan blood pressure medications, which were contaminated with NDMA. New Yorkers Elizabeth and John Duffy filed the lawsuit on behalf of those who bought the tainted blood pressure drugs. These drugs were recalled in July and August of 2018, although it is believed the contamination could have been occurring since 2012. The lawsuit, filed in federal court in New York, names four companies as defendants in the suit—two who distributed valsartan and two pharmacies that sold the contaminated drug.
The lawsuit claims these companies were negligent, while also committing fraud, and manufacturing a defective product. Companies named in the valsartan class action include Solco Healthcare, Prinston Pharmaceutical, Inc., Walgreens, and Throggs Neck Pharmacy. Both John and Elizabeth Duffy claim they took contaminated versions of valsartan for a significant period of time.
Getting the Help You Need After Taking a Recalled Blood Pressure Drug
 When we are prescribed drugs for a medical condition by our physicians, we believe the drug is safe. When that turns out not to be the case, your client could need assistance. Your client’s ability to receive treatment for their injuries related to a tainted blood pressure drug may be limited. However, help can come from USClaims. At USClaims, pre-settlement funding can help your clients pay those unexpected expenses (including medical expenses and lost wages) in anticipation of a court judgment or settlement. Call 1-877-USCLAIMS today for the information you and your clients need and deserve.
When we are prescribed drugs for a medical condition by our physicians, we believe the drug is safe. When that turns out not to be the case, your client could need assistance. Your client’s ability to receive treatment for their injuries related to a tainted blood pressure drug may be limited. However, help can come from USClaims. At USClaims, pre-settlement funding can help your clients pay those unexpected expenses (including medical expenses and lost wages) in anticipation of a court judgment or settlement. Call 1-877-USCLAIMS today for the information you and your clients need and deserve.